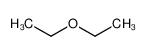
| Product name | Diethyl ether |
|---|
| Product number | - |
|---|---|
| Other names | Ethane, 1,1‘-oxybis- |
| Identified uses | For industry use only. Food Additives: EXTRACTION_SOLVENT |
|---|---|
| Uses advised against | no data available |
| Company | MOLBASE (Shanghai) Biotechnology Co., Ltd. |
|---|---|
| Address | Floor 4 & 5, Building 12, No. 1001 North Qinzhou Road, Xuhui District, Shanghai, China |
| Telephone | +86(21)64956998 |
| Fax | +86(21)54365166 |
| Emergency phone number | +86-400-6021-666 |
|---|---|
| Service hours | Monday to Friday, 9am-5pm (Standard time zone: UTC/GMT +8 hours). |
Flammable liquids, Category 1
Acute toxicity - Oral, Category 4
Specific target organ toxicity – single exposure, Category 3
2.2 GHS label elements, including precautionary statements| Pictogram(s) |   |
|---|---|
| Signal word | Danger |
| Hazard statement(s) | H224 Extremely flammable liquid and vapour H302 Harmful if swallowed H336 May cause drowsiness or dizziness |
| Precautionary statement(s) | |
| Prevention | P210 Keep away from heat, hot surfaces, sparks, open flames and other ignition sources. No smoking. P233 Keep container tightly closed. P240 Ground and bond container and receiving equipment. P241 Use explosion-proof [electrical/ventilating/lighting/...] equipment. P242 Use non-sparking tools. P243 Take action to prevent static discharges. P280 Wear protective gloves/protective clothing/eye protection/face protection. P264 Wash ... thoroughly after handling. P270 Do not eat, drink or smoke when using this product. P261 Avoid breathing dust/fume/gas/mist/vapours/spray. P271 Use only outdoors or in a well-ventilated area. |
| Response | P303+P361+P353 IF ON SKIN (or hair): Take off immediately all contaminated clothing. Rinse skin with water [or shower]. P370+P378 In case of fire: Use ... to extinguish. P301+P312 IF SWALLOWED: Call a POISON CENTER/doctor/…if you feel unwell. P330 Rinse mouth. P304+P340 IF INHALED: Remove person to fresh air and keep comfortable for breathing. P312 Call a POISON CENTER/doctor/…if you feel unwell. |
| Storage | P403+P235 Store in a well-ventilated place. Keep cool. P403+P233 Store in a well-ventilated place. Keep container tightly closed. P405 Store locked up. |
| Disposal | P501 Dispose of contents/container to ... |
none
3.Composition/information on ingredients 3.1 Substances| Chemical name | Common names and synonyms | CAS number | EC number | Concentration |
|---|---|---|---|---|
| Diethyl ether | Diethyl ether | 60-29-7 | none | 100% |
Consult a physician. Show this safety data sheet to the doctor in attendance.
If inhaledFresh air, rest. Artificial respiration may be needed. Refer for medical attention.
In case of skin contactRemove contaminated clothes. Rinse skin with plenty of water or shower.
In case of eye contactFirst rinse with plenty of water for several minutes (remove contact lenses if easily possible), then refer for medical attention.
If swallowedRinse mouth. Do NOT induce vomiting. Give one or two glasses of water to drink. Refer for medical attention .
4.2 Most important symptoms/effects, acute and delayedVapor inhalation may cause headache, nausea, vomiting, and loss of consciousness. Contact with eyes will be irritating. Skin contact from clothing wet with the chemical may cause burns. (USCG, 1999)
4.3 Indication of immediate medical attention and special treatment needed, if necessaryINHALATION: Cough. Sore throat. Drowsiness. Vomiting. Headache. Labored breathing. Unconsciousness. First aid: Fresh air, rest. Artificial respiration may be needed. Refer for medical attention. SKIN: Symptoms: Dry skin. First aid: Remove contaminated clothes. Rinse skin with plenty of water or shower. EYES: Symptoms: Redness. Pain. First aid: First rinse with plenty of water for several minutes (remove contact lenses if easily possible), then take to a doctor. INGESTION: Symptoms: Dizziness. Drowsiness. Vomiting. First aid: Rinse mouth. Do NOT induce vomiting. Give plenty of water to drink. Refer for medical attention.
5.Fire-fighting measures 5.1 Extinguishing media Suitable extinguishing mediaWater may be ineffective ... But water should be used to keep fire-exposed containers cool. Use water spray dry chemical, foam, or carbon dioxide.
5.2 Specific hazards arising from the chemicalBehavior in Fire: Vapor is heavier than air and may travel considerable distance to a source of ignition and flash back. Decomposes violently when heated. (USCG, 1999)
5.3 Special protective actions for fire-fightersWear self-contained breathing apparatus for firefighting if necessary.
6.Accidental release measures 6.1 Personal precautions, protective equipment and emergency proceduresUse personal protective equipment. Avoid dust formation. Avoid breathing vapours, mist or gas. Ensure adequate ventilation. Evacuate personnel to safe areas. Avoid breathing dust. For personal protection see section 8.
6.2 Environmental precautionsRemove all ignition sources. Evacuate danger area! Consult an expert! Personal protection: filter respirator for organic gases and vapours adapted to the airborne concentration of the substance. Collect leaking liquid in sealable containers. Absorb remaining liquid in sand or inert absorbent. Then store and dispose of according to local regulations.
6.3 Methods and materials for containment and cleaning upSpills and leakage: Absorb with paper. Evaporate completely all spilt surface. Dispose by burning the paper after complete ventilation of vapor.
7.Handling and storage 7.1 Precautions for safe handlingAvoid contact with skin and eyes. Avoid formation of dust and aerosols. Avoid exposure - obtain special instructions before use.Provide appropriate exhaust ventilation at places where dust is formed. For precautions see section 2.2.
7.2 Conditions for safe storage, including any incompatibilitiesFireproof. Separated from strong oxidants. See Chemical Dangers. Cool. Keep in the dark. Store only if stabilized.Separate from oxidizing materials. Store in a cool, dry, well-ventilated area. Avoid sunlight.
8.Exposure controls/personal protection 8.1 Control parameters Occupational Exposure limit valuesNIOSH questioned whether the PEL proposed by OSHA for ethyl ether [TWA 400 ppm; STEL 500 ppm] was adequate to protect workers from recognized health hazards.
Biological limit valuesno data available
8.2 Appropriate engineering controlsHandle in accordance with good industrial hygiene and safety practice. Wash hands before breaks and at the end of workday.
8.3 Individual protection measures, such as personal protective equipment (PPE) Eye/face protectionSafety glasses with side-shields conforming to EN166. Use equipment for eye protection tested and approved under appropriate government standards such as NIOSH (US) or EN 166(EU).
Skin protectionWear impervious clothing. The type of protective equipment must be selected according to the concentration and amount of the dangerous substance at the specific workplace. Handle with gloves. Gloves must be inspected prior to use. Use proper glove removal technique(without touching glove's outer surface) to avoid skin contact with this product. Dispose of contaminated gloves after use in accordance with applicable laws and good laboratory practices. Wash and dry hands. The selected protective gloves have to satisfy the specifications of EU Directive 89/686/EEC and the standard EN 374 derived from it.
Respiratory protectionWear dust mask when handling large quantities.
Thermal hazardsno data available
9.Physical and chemical properties| Physical state | Clear liquid |
|---|---|
| Colour | Colorless, volatile, mobile liquid |
| Odour | Sweetish, pungent odor |
| Melting point/ freezing point | -116ºC |
| Boiling point or initial boiling point and boiling range | 34.6°C(lit.) |
| Flammability | Class IA Flammable Liquid: Fl.P. below 22.78°C and BP below 37.78°C.Extremely flammable. |
| Lower and upper explosion limit / flammability limit | Lower flammable limit: 1.9% by volume; Upper flammable limit: 36.0% by volume |
| Flash point | -40°C |
| Auto-ignition temperature | 160°C |
| Decomposition temperature | no data available |
| pH | no data available |
| Kinematic viscosity | 0.2448 centipoise at 20°C |
| Solubility | In water:69 g/L (20 ºC) |
| Partition coefficient n-octanol/water (log value) | log Kow= 0.89 |
| Vapour pressure | 28.69 psi ( 55 °C) |
| Density and/or relative density | 0.706g/mLat 25°C(lit.) |
| Relative vapour density | 2.6 (vs air) |
| Particle characteristics | no data available |
no data available
10.2 Chemical stabilityIt is slowly oxidized by action of air, moisture, and light, with formation of peroxides.
10.3 Possibility of hazardous reactionsWhen shaken under absolutely dry conditions ether can generate enough static electricity to start a fire.The vapour is heavier than air and may travel along the ground; distant ignition possible. As a result of flow, agitation, etc., electrostatic charges can be generated.Occasional explosions have occurred when aluminum hydride was stored in ether. The explosions have been blamed on the presence of carbon dioxide impurity in the ether, [J. Amer. Chem. Soc. 70:877(1948)]. Diethyl ether and chromium trioxide react violently at room temperature. Solid acetyl peroxide in contact with ether or any volatile solvent may explode violently. A 5-gram portion in ether detonated while being carried, [Chem. Eng. News 27:175(1949)]. Nitrosyl perchlorate ignites and explodes with diethyl ether. A mixture of ether and ozone forms aldehyde and acetic acid and a heavy liquid, ethyl peroxide, an explosive, [Mellor 1:911(1946-1947)].
10.4 Conditions to avoidno data available
10.5 Incompatible materialsBoron triazide, bromine trifluoride, bromine pentafluoride, bromine, iodine heptafluoride, silver perchlorate, fluorine nitrate, permanganic acid, nitric acid, hydrogen peroxide, peroxodisulfuric acid, iodine(VII) oxide, peat soils, thiotriazyl perchlorate, sulfonyl chloride, sulfur, uranyl nitrate, and wood pulp extracts.
10.6 Hazardous decomposition productsWhen heated to decomposition it emits acrid smoke and irritating fumes.
11.Toxicological information Acute toxicity- Oral: LD50 Rat oral 3.56 (3.23 - 3.92 g/kg in a single vehicle) /From table/
- Inhalation: LC50 Mouse inhalation 186 mg/L/90 min
- Dermal: LD50 Rabbit percutaneous 20 mL/kg. /From table/
no data available
Serious eye damage/irritationno data available
Respiratory or skin sensitizationno data available
Germ cell mutagenicityno data available
Carcinogenicityno data available
Reproductive toxicityno data available
STOT-single exposureno data available
STOT-repeated exposureno data available
Aspiration hazardno data available
12.Ecological information 12.1 Toxicity- Toxicity to fish: LC50 Pimephales promelas (fathead minnow, age 29 days) 2560 mg/L/96 hr; flow-through, 24.8°C, pH 7.76, dissolved oxygen 7.1 mg/L, hardness 45.1 mg/L CaCO3, alkalinity 41.5 mg/L CaCO3
- Toxicity to daphnia and other aquatic invertebrates: EC50; Species: Daphnia magna (Water flea); Conditions: freshwater, static, pH 8.0; Concentration: 165 mg/L for 24 hr; Effect: behavior, equilibrium
- Toxicity to algae: no data available
- Toxicity to microorganisms: no data available
AEROBIC: Diethyl ether has been included in a list of compounds which were not biodegraded in a relatively short time either in screening tests which utilized sewage sludge inocula or soil inocula(1). Many ethers are known to be resistant to biodegradation(1). The 5-day biological oxygen demand measured for diethyl ether in screening tests ranged from 0%(2,3) to approximately 1.1%(2) in studies using the standard dilution technique with sewage inocula and 0% in a screening study using an activated sludge inoculum(4). A lag time of >10 days was observed in the latter study with activated sludge(4). In a study which used the seawater dilution method with a sewage inoculum, 0% theoretical BOD was observed over a 5-day period(3). No change was observed in the biological oxygen demand in tests of a semi-continuous activated sludge biological treatment simulator which tested for the removal of diethyl ether at a concn of 200 to 800 ppm in a domestic sewage feed over a period of 24 hours(5). A biodegradation study of diethyl ether, based on TOC measurements (GC analysis), using an activated sludge seed and an initial diethyl ether concn of 100 mg/L, indicated 2.5% (6.5%) biodegradation over a period of 4 weeks(6).
12.3 Bioaccumulative potentialBCF values of 0.9 to 1.4 and <1.7 to 9.1 were measured for carp exposed to 500 and 50 ug/L of diethyl ether over the course of a 6 week incubation period(1). According to a classification scheme(2), these BCFs suggest bioconcentration in aquatic organisms is low.
12.4 Mobility in soilThe Koc of diethyl ether is estimated as approximately 73(SRC), using a log Kow of 0.89(1) and a regression-derived equation(2). According to a classification scheme(3), this estimated Koc value suggests that diethyl ether is expected to have high mobility in soil(SRC). A vapor-phase partition coefficient of 15.49 was measured for diethyl ether at 30°C in the silt and clay size fraction of a calcareous soil (1.2% organic carbon w/w) from Southern Nevada at 52% relative humidity(4).
12.5 Other adverse effectsno data available
13.Disposal considerations 13.1 Disposal methods ProductThe material can be disposed of by removal to a licensed chemical destruction plant or by controlled incineration with flue gas scrubbing. Do not contaminate water, foodstuffs, feed or seed by storage or disposal. Do not discharge to sewer systems.
Contaminated packagingContainers can be triply rinsed (or equivalent) and offered for recycling or reconditioning. Alternatively, the packaging can be punctured to make it unusable for other purposes and then be disposed of in a sanitary landfill. Controlled incineration with flue gas scrubbing is possible for combustible packaging materials.
14.Transport information 14.1 UN Number| ADR/RID: UN1155 | IMDG: UN1155 | IATA: UN1155 |
| ADR/RID: DIETHYL ETHER (ETHYL ETHER) |
| IMDG: DIETHYL ETHER (ETHYL ETHER) |
| IATA: DIETHYL ETHER (ETHYL ETHER) |
| ADR/RID: 8 | IMDG: 8 | IATA: 8 |
| ADR/RID: I | IMDG: I | IATA: I |
| ADR/RID: no | IMDG: no | IATA: no |
no data available
14.7 Transport in bulk according to Annex II of MARPOL 73/78 and the IBC Codeno data available
15.Regulatory information 15.1 Safety, health and environmental regulations specific for the product in question| Chemical name | Common names and synonyms | CAS number | EC number |
|---|---|---|---|
| Diethyl ether | Diethyl ether | 60-29-7 | none |
| European Inventory of Existing Commercial Chemical Substances (EINECS) | Listed. | ||
| EC Inventory | Listed. | ||
| United States Toxic Substances Control Act (TSCA) Inventory | Listed. | ||
| China Catalog of Hazardous chemicals 2015 | Listed. | ||
| New Zealand Inventory of Chemicals (NZIoC) | Listed. | ||
| Philippines Inventory of Chemicals and Chemical Substances (PICCS) | Listed. | ||
| Vietnam National Chemical Inventory | Listed. | ||
| Chinese Chemical Inventory of Existing Chemical Substances (China IECSC) | Listed. | ||
| Creation Date | Aug 13, 2017 |
|---|---|
| Revision Date | Aug 13, 2017 |
- CAS: Chemical Abstracts Service
- ADR: European Agreement concerning the International Carriage of Dangerous Goods by Road
- RID: Regulation concerning the International Carriage of Dangerous Goods by Rail
- IMDG: International Maritime Dangerous Goods
- IATA: International Air Transportation Association
- TWA: Time Weighted Average
- STEL: Short term exposure limit
- LC50: Lethal Concentration 50%
- LD50: Lethal Dose 50%
- EC50: Effective Concentration 50%
- IPCS - The International Chemical Safety Cards (ICSC), website: http://www.ilo.org/dyn/icsc/showcard.home
- HSDB - Hazardous Substances Data Bank, website: https://toxnet.nlm.nih.gov/newtoxnet/hsdb.htm
- IARC - International Agency for Research on Cancer, website: http://www.iarc.fr/
- eChemPortal - The Global Portal to Information on Chemical Substances by OECD, website: http://www.echemportal.org/echemportal/index?pageID=0&request_locale=en
- CAMEO Chemicals, website: http://cameochemicals.noaa.gov/search/simple
- ChemIDplus, website: http://chem.sis.nlm.nih.gov/chemidplus/chemidlite.jsp
- ERG - Emergency Response Guidebook by U.S. Department of Transportation, website: http://www.phmsa.dot.gov/hazmat/library/erg
- Germany GESTIS-database on hazard substance, website: http://www.dguv.de/ifa/gestis/gestis-stoffdatenbank/index-2.jsp
- ECHA - European Chemicals Agency, website: https://echa.europa.eu/